For example, some of the violent reactions demonstrated the necessity of checking labels and being careful while mixing chemicals to prevent dangerous reactions. Ramifications of this experiment are broad. This experiment verifies that certain electron configurations are more likely to react than others. 
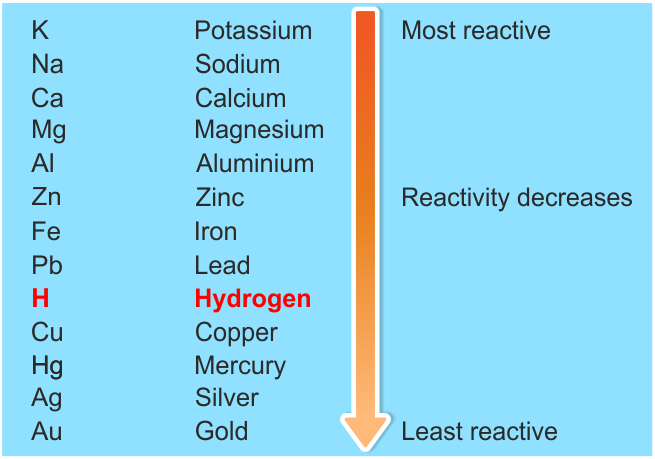
The structure of the atom is responsible for the electron configuration. Valence electrons determine the charge of the ion and the ionization energy quantity. Different electron configurations, when combined with different ionization energies as required, produce ions of different charges. The theory associated with this experiment is the atomic theory, more specifically electron configuration. Ample time must be allowed for the reaction to occur. Though a reaction is occurring, an untrained eye may disregard the signs of a reaction. Some reactions take several minutes to take place, and others produce only slight bubbling. A major human source of error is ending a reaction too soon. This causes discrepancies between what should have happened in a given reaction and what was observed. The oxide coating on some of the metals, such as calcium, may have delayed or prevented reactions from occurring. For example, if any of the samples of ionic solutions were contaminated, then the reaction would have been altered by possibly entering another ion into the reaction. Some errors were human errors and were the result of lack of experience. Elements to the left will replace elements to the right.ĭiscussion: There were several possible sources of error in this experiment. It is ordered from most reactive element to least reactive element. This activity series indicates which elements will replace other elements in single replacement (metathesis) reactions. Net ionic equations remove spectator ions from the chemical equations. Only reactions that occurred are listed below. This table outlines the reactions that took place as reported in Observations Part B. Figure 15.1 ( view | download) is a partial periodic table. The periodic table reveals a great deal of information about the elements based on the placement of elemental symbols. The columns of the periodic table contain elements with similar properties. The periodic table is arranged according to the number of protons in one atom of each element. The periodic table was developed in the 1860s by Dmitri Mendeleev. All common metals are solids at room temperature, except for mercury, which is a liquid. Some of the physical properties of metals include high thermal and electrical conductivity, high luster, malleability, and ductility. You need these materials: 0.2 M Ca(NO 3) 2, 0.2 M Mg(NO 3) 2, 0.2 M Zn(NO 3) 2, 0.2 M Fe(NO 3) 3, 0.2 M FeSO 4, 0.2 M SnCl 4, 0.2 M Cu(NO 3) 2, 6 M HCl, 7 small pieces each of calcium, magnesium, zinc, iron, tin, and copperĬhemical elements are usually classified by their properties into three groups: metals, nonmetals, and metalloids. You need this equipment: 7 small test tubes, test tube rack Let's become familiar with the relative activities of metals in chemicals! 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
